Glass for Electronics
|
|
This "Glass for Electronics" article in a 1965 issue of Electronics World magazine provides some really interesting information about the properties of glass which I, for one, either never knew or have forgotten. One such point is that glass is typically defined with a "softening" temperature rather than a melting temperature. That is because the final characteristics of the glass is highly dependent on the cooling down time/temperature profile. Those of us having been in the world of automated printed circuit assembly (PCA) solder oven operation are familiar with the criticality of time/temperature profiles, so the concept is not new. In the case of PCA's, profiling is necessary to accommodate the often widely varying thermal dimension changes over temperature to prevent fracturing. With glass, it is the final atomic alignment (or misalignment) that is dependent on the cooling process, akin to tempering of metal (although the metal is not heated to the point of liquidus flow). At the time of this article, Corning Glass Works claimed to have >100k unique formulas for glass using various mixes of elements. Glass for Electronics
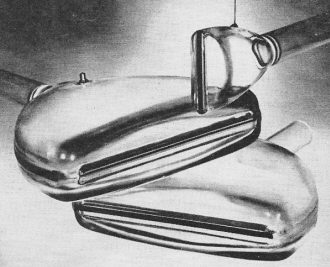
New types of glass are playing an increasingly important role in electronics. They are finding use in delay lines, precision resistors and capacitors, radomes, and CRT's used for readout. From the time of the first pioneer efforts in electronics, glass has been prominent as an insulator, an enclosure, and a dielectric. Special glasses created in recent years have provided greatly improved electronic products and, in many instances, have made simplified manufacturing techniques possible. The variety of glasses now available is staggering. Practically every element has been utilized in glass-making, and some glasses contain 20 to 30 distinct ingredients. Corning Glass Works alone reports that it has more than 100,000 different glass formulas. Fig. 1 - Delay lines used in radar, computers, and other electronic devices can be made from precise polygon of hard glass. Fig. 2 - High-temperature, high-strength glass-ceramics are used as combination nose cone/radome in many guided missiles. Despite this variety, about 90% of all glass produced in the United States is a type called "soda lime," which is made from approximately 70% silica sand (silicon oxide) mixed with oxides of sodium and calcium. The alkali oxides act as fluxing agents and form, with the silica, a mixture that softens and flows at a lower temperature than pure silica. As the glass cools from the temperature at which it is a true fluid, its viscosity increases rapidly. The resulting decrease in atomic mobility is sufficient to prevent the formation of an orderly atomic pattern, and normally glass does not form a crystalline structure. Instead, a random pattern is frozen into the glass, a fact which accounts for many of its properties. Since the transition from a solid to a liquid takes place over a temperature range, glass has no definite melting point. It is customary, therefore, to refer to the softening temperature of glass rather than its melting point. Soda-lime glass is widely used for windows, bottles, drinking glasses, and lamp bulbs, but its applications in electronics are limited. With a relatively low softening temperature, it cannot be used where excessive heat is encountered. Surface conductivity, an important consideration for many applications, is low because glass with high alkali content tends to absorb moisture. under conditions of high humidity, the resistivity of soda-lime glass is only about 107 ohms/square, compared with 1010 ohms/square for glass with low alkali content. This restricts its electronic usage. Hard Glass The terms "hard" and "soft" as applied to glass refer to service temperature rather than mechanical hardness, the dividing point being about 400°C. Aluminosilicate glass, which has low sodium content and contains a high proportion of aluminum oxide, has a softening temperature over 900°C and will give useful service up to 650°C. It has good electrical and chemical properties and is often used as an envelope for high-performance power tubes and traveling-wave tubes. When melted in optical quality, it is virtually free of defects and is then useful for faceplates for cathode-ray tubes. The hardest glasses are those containing the highest percentage of silica. A glass of about 96% silica is made by chemically removing almost all elements except silica from boro-silicate glass. This leaves a porous structure, so it is then necessary to fire the glass again - a process that adds to the expense. Nevertheless, such glass does not begin to soften below 1000°C and can be used regularly at 800°C. It can be heated to a cherry red, then plunged into ice water without ill effect. Glass of this kind has excellent infrared transmission properties and has been used for windows in heat-seeking missiles. An even harder glass is made by fusing pure silica in which impurity levels are held to less than one part-per-million. This product is the most transparent glass ever made and is used in the finest telescopes. It is also used for laser-beam mirrors and for infrared and ultraviolet windows. Because it has a service temperature from 900 to l100°C, it is used for crucibles needed to grow silicon crystals for diodes and transistors. Fused silica glass will transmit an ultrasonic signal with practically no attenuation or scattering, and this has led to its use in ultrasonic delay lines for radars and computers. The electronic signal is converted by a transducer into an ultrasonic signal which travels through the glass until it is picked up by a second transducer and converted back into electrical energy. Relatively long delay times - 150 micro-seconds or more - are achieved by means of a folded path whereby the energy is reflected from facet to facet of a precisely ground polygon. Delay times up to several thousand microseconds are thus possible in a relatively small physical space. The advantages of very low acoustical attenuation are offset where temperature varies widely by the fact that the coefficient of time delay in fused silica amounts to about 80 PPM/°C. To eliminate the need for temperature control, a special glass was developed with a coefficient of time delay of only 0.5 PPM/°C. This material was used in the delay lines (Fig. 1) in the shift registers of the digital guidance computer for the Gemini manned spacecraft. An important consideration in the selection of this glass was its insensitivity to vibration and other mechanical forces encountered in space flight. Glass-Ceramics Even tougher than fused silica glass is glass-ceramic, which is so hard that it can be used to make ball bearings for machinery. Glass-ceramic devices are first formed by ordinary techniques from a special glass batch to which nucleating agents have been added to promote crystal growth. After the device has been cooled, it is reheated and subjected to treatment that causes the crystallization of billions of invisible crystallites throughout the glass body. Glass-ceramic produced by heat treatment is opaque and far stronger and harder than the parent glass. It has greater impact and abrasion resistance and improved thermal and electrical properties. Types have been developed with excel-lent dielectric properties at microwave frequencies, and these have proved useful for missile radomes. A radome (Fig. 2) permits passage of radio waves for guidance of the missile and at the same time protects the internal guidance system from its environment. Such glass-ceramic radomes have nearly constant dielectric properties over a range from 25 to 500°C; the dissipation and loss factors are nominal. Photosensitive Glass Fig. 3 - Glass can be fabricated in a number of intricate patterns as shown by this arrangement of fluid amplifier components. Fig. 4 - These precision quarter-watt metal-oxide film resistors use glass bases and can dissipate one-tenth of a watt. One of the most interesting and useful recent developments is a type of glass that is sensitive to ultraviolet light. The discovery grew out of the observation that the windows of old houses sometimes take on a violet coloration. Further investigation led to the finding that certain types of glass, under the influence of ultraviolet radiation, form nucleated crystals. Finally, it was determined that such crystals could be further developed by heat treatment and that they are then 20 times more susceptible to attack by hydrofluoric acid than glass that has not been exposed to such radiation. When exposed to ultraviolet light and heat, photosensitive glass behaves much like photographic film. An image is formed that is a permanent part of the glass and which extends in depth throughout the body of the glass. Exposed areas turn an opalescent white after heat treatment, while unexposed areas remain clear. Because of the marked difference in sensitivity of the exposed portion, the pattern produced by photographic means can be etched out with hydrofluoric acid, leaving an exact copy of the negative through which the exposure was made. The process of chemically etching or machining glass has proved a most useful discovery, since glass is difficult to machine by conventional techniques. Fig. 3 shows how photosensitive glass can be chemically machined to form the intricate pattern needed for a fluid amplifier. Developed by the Diamond Ordnance Fuze Laboratories, these devices utilize fluids to perform logic functions normally accomplished with electronic circuits. Other applications for photosensitive-glass etching include printed-circuit boards, solid-state substrates, optical coding discs, and various laminated structures. Fine glass screens, containing more than 350,000 precisely located holes per square inch, are utilized for aperture masks in the manufacture of color-television picture tubes. Coated Glass Glass itself is an excellent insulator, but it can be made to conduct electricity by firing a metallic-oxide film onto its surface. Although such films are usually less than 0.1 mil thick, they are durable, stable, and-in most cases - transparent. Through selection of the metallic oxides to be used and control of thickness, electrical resistance may be obtained anywhere in a range from about 10 to 106 ohms/square. Coated glass was initially used for residential and industrial heating panels, self-defrosting windshields and rear-view mirrors, and similar devices. Since the metallic surface reflects about 50% of the infrared radiation which strikes it, while letting most of the visible light through, coated glass is also used for windows that permit viewing of industrial processes involving great heat without discomfort to the viewer. If coated glass panels are grounded, they can be used to shield delicate electronic instruments against r.f. radiation. Used in fluorescent lighting fixtures, they ground interference but will permit light rays to pass relatively unobstructed. Tin oxide fired on a glass substrate that matches its coefficient of expansion forms the basis for a tough resistor with excellent stability under adverse conditions. Such resistors may vary from tiny devices, shown in Fig. 4, to massive units four feet long and five inches in diameter which are used as dummy antenna loads for testing transmitters. They have low noise characteristics and may be hermetically sealed in glass envelopes for protection against moisture and contaminants. A related application of the same technique is the production of inductors by firing metalized silver conductors into special glass coil forms. The thin metal coatings have almost zero turn-to-turn capacitance, and the inductors can be used at frequencies to 250 mc. (Fig. 5). Conducting Mosaics
Fig. 5 - Precision variable inductors are fired on glass bases. Fig. 6 - Cathode-ray tubes with conducting mosaic faceplates are often used in high-speed electrostatic printing equipment. Mosaics consisting of a large number of microminiature conductors, precisely aligned and hermetically sealed in glass, are now being produced, and this has led to the development of a system of electronic printing. A typical array consists of conductors one mil in diameter spaced at intervals of four mils, providing a density of 62,500 conductors per square inch. The mosaics are sealed into the face of cathode-ray tubes. Some of the tubes presently available are shown in Fig.6. One of the first applications of conducting mosaics was for the printing of address labels for magazines. Television-type signals were fed to the cathode-ray tube by a character generator, and electrostatic patterns of the characters were formed on a continuous paper web as it passed before the tube. The charged paper was first processed to make the images permanent and was then cut into individual labels. This system made it possible to print 36 labels each second. Similar tubes are used in an arrangement for keeping track of individual railroad cars. As trains enter a yard and pass by a scanner, images are taken sequentially of each car in each train, The car numbers showing in the electrostatic print-outs enable fast pinpointing of the location of any car of any train in the yard. Cathode-ray tubes with conducting mosaics are also used in certain facsimile systems. Glass for Capacitors Although serviceable capacitors were made many years ago from window glass, it has since been possible to improve the product considerably by careful selection of the best glass for the dielectric. A number of factors enter into the choice. Pure silica glass has the lowest dielectric constant - about 3.8. The dielectric constant is increased by the addition of fluxing agents to the glass mixture, the largest increases resulting from the heavier ions. Glass containing a high percentage of lead, for example, has a dielectric constant of 15 to 17. However, such glass has high power factor and loss and is therefore not used for capacitors. Even more important than dielectric constant is dielectric strength. This follows from the fact that the amount of energy that can be stored in a capacitor varies in direct proportion. to the dielectric constant, but also in proportion to the square of the dielectric strength. Therefore, a glass with twice the dielectric strength is as effective as one with four times the dielectric constant. It has already been pointed out that soda-lime glass has relatively low resistivity and hence would not be the best choice for capacitors. Instead, a potash-lead glass is often used. It has a dielectric constant of about 8.8, high dielectric strength, and low power factor and loss. It can be drawn in the form of a thin, flexible ribbon about one mil thick, free from holes, cracks, or other imperfections. Furthermore, it matches the coefficient of expansion of the aluminum foil to which it is mated. Alternate layers of ribbon and aluminum foil are sealed together at high temperature to form rugged, monolithic units. Leads are attached, and the dielectric stack is then fused into a compatible glass case. Fixed glass capacitors are produced in a range of values from about 0.5 pf. to about 150 μf. and in working voltages to 6000 volts. They are characterized by high "Q," good stability, and excellent efficiency at temperatures to 300°C. Miniature glass trimmer capacitors (Fig. 7) provide a capacitance change of only 0.4 pf. per turn, permitting very precise tuning. The tuning curve is linear, and the units can be hermetically sealed against moisture. A very large number of special glasses have been devised to fill particular needs. For instance, rods made of borate- or silicate-base optical glass and doped with oxides of the rare-earth elements neodymium or ytterbium now serve as laser elements. The glass must be of the highest quality, and the rods themselves are tested with a gas laser source to assure optical homogeneity as well as end flatness and parallelism. Eastman Kodak Company makes such glass laser rods in sizes up to one inch in diameter and 36 inches in length. Power output as great as three joules per cubic centimeter of glass is reported. High-silica glass is manufactured in a porous form containing billions of microscopic holes. This product is useful as a moisture-getter, and small slabs of it may be incorporated in semiconductor cases and relay envelopes for that purpose. The glass is mechanically strong, non-dusting, and non-flaking. Porous glass is also fabricated in the form of semi-permeable membranes for isotope separation and is used for chromatography and diffusion studies. A non-porous glass membrane is used in the sensing electrode in a pH-measuring instrument, which determines the acidity or alkalinity of a solution by means of the hydrogen-ion concentration. A solution of known acidity is placed in the glass electrode and, when the electrode is immersed in the unknown solution, a voltage develops across the glass membrane dependent upon the difference in the hydrogen-ion concentration of the two solutions. Since the membrane is not porous, there is no transfer of electrons at the electrode-solution interface. However, special glass is needed to resist the effects of strong acid or alkaline solutions. Glass to resist nuclear radiation is usually made with high lead content. It is cast in massive slabs for use in shielding windows in radiation laboratories, aboard nuclear vessels, and in hospitals where radioactive materials are employed. Because of its inorganic nature and its random structure, glass offers high resistance to x-ray and gamma radiation and will not darken with use. Fig. 7. Precision hermetically sealed glass trimmer capacitors are moisture-proof and possess linear tuning curves. A related product is gamma-ray-sensitive glass, a specially developed glass that will fluoresce upon exposure to ultraviolet light after irradiation by gamma rays. The intensity of the light emission is proportional to the total gamma-ray dose received, so badges made from this material serve as sensitive dosimeters. Dosages from 5 to 1000 roentgens can be measured in this way.
Posted November 28, 2022 |
|

 By John R. Collins
By John R. Collins