|
|
|||||||||
| Software: RF Cascade Workbook | RF Symbols for Office | RF Symbols & Stencils for Visio | Espresso Workbook | ||||||||||
|
|||||||||||||||||||||||||||||||
   |
   |
||||||||||||||||||||||||||||||
|
Please Support RF Cafe by purchasing my ridiculously low-priced products, all of which I created. RF & Electronics Symbols for Visio RF & Electronics Symbols for Office RF & Electronics Stencils for Visio T-Shirts, Mugs, Cups, Ball Caps, Mouse Pads These Are Available for Free |
|||||||||||||||||||||||||||||||

Fundamentals of Color TV
March 1954 Radio &
Television News
| March 1954 Radio & TV News |
 [Table of Contents] [Table of Contents]
Wax nostalgic about and learn from the history of early electronics. See articles from Radio & Television News, published 1919-1959. All copyrights hereby acknowledged. |
Quite a few articles on color television were published in trade and hobby magazines in the 1950s and 1960s as the technology was adopted and fine tuned. The electronic circuitry aspect of transmitting and receiving chromaticity, intensity, synchronization, and audio was impressive, but the science that went into color research was equally amazing. As with so many things we take for granted because someone else did all the hard work of figuring out how to make something work and then making it available to us at an affordable price, the physics of human color perception needed intense study in order to produce a pleasing image on the cathode ray tube (CRT). The key to understanding color is the chromaticity diagram, based in the human tristimulus color space, which is described in detail herein. It concludes with a triangle superimposed on the chromaticity diagram indicating the region occupied by the color range available for a given CRT based on phosphor type and electron gun energy.
Part 1. Color fundamentals and their application in color television for radio and TV service technicians.
 By Milton S. Kiver
By Milton S. Kiver
Pres. Television Communications Institute
Color forms one of the most intimate contacts in our everyday life - we wear colored clothes, we use colored objects, we live in colored houses, and we eat colored food. Yet, in spite of this close contact with color, most people have only a casual knowledge of the nature of color or of color mixing. To the television technician, color possesses added significance because of its application in color television. Such terms as color primaries, hue, saturation, chromaticity, and luminance will be commonly used in any description of a color television receiver, both from the standpoint of operation and of service. What do these words mean? How do they tie in with color television? These are some of the questions the service technician will be confronted with and now, while the art is still young, is as good a time as any to learn about them.
Editor's Note
Although color television is a commercial reality, few TV service technicians will have the opportunity to install or work with sets for some time to come. In the meantime, the service technician can ground himself thoroughly on the fundamentals of the subject so as to be prepared when color TV sets become widely available. This article is the first of a series that will attempt to present all the background necessary for the servicing of color TV receivers. Actual schematic diagrams of color TV sets will be analyzed when they become available and once the fundamentals of color TV have been thoroughly covered. Of course, other articles in Radio & Television News will present the latest advances in color TV as they occur.
Let us start off with color primaries. Anyone who has ever experimented with projector lamps has discovered that when different colored lights from several projectors are combined, the resultant color seen by an observer will differ from the color of any of the projected beams. Thus, for example, yellow can be formed by combining red and green light; white light can be produced by combining red, green, and blue. The color of the light formed will appear to the eye as a complete color and the eye will be unable to distinguish the various components of the mixture that united to form the new color.

Fig. 1 - (A) A new color is formed between color "A" and color "B" as a result of mixing color "A" and color "B". (B) Adding three original colors, "A", "B", and "C", results in four new ones.
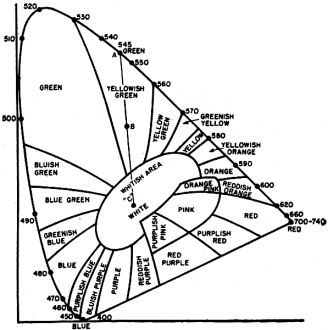
Fig. 2 - A chromaticity diagram. See text for details.
Fig. 3 - The line drawn between points "R" and "G" passes through all the colors that can be obtained by mixing these two shades of the red and green hues.
Fig. 4 - When moving from "A" to "C," the green becomes less and less saturated, or lighter in intensity.
Fig. 5 - Colors "F" and "G" will produce white when mixed in proper proportions; they are complementary.
Fig. 6 - Within the triangle are the colors that can be seen on a color TV screen.
This method of color formation is illustrated in. Fig. 1A. Two circles of colored light are projected onto a screen and positioned so that they overlap to some extent. Within the overlapping region, a new color will be produced by the addition of color "A" and color "B." Where the circles of light do not overlap, each light will retain its original color. If a third circle of light is added, as shown in Fig. 1B, then a maximum of seven colors can be obtained. These would be: color "A" color "B" color "C" color "D" (formed from "A" and "B"), color "E" (formed from "A" and "C"), color "F" (formed from "B" and "C"), and color "G" (formed from "A," "B," and "C") - and each would differ from the other. In the areas where the circles of light overlapped, the eye would not be able to distinguish each of the colors forming the mixture, but instead would see the final color produced. Thus, color "A" and "B" would not appear to the eye as color "A" and color "B," but as some new color which we can call color "D." The same would be true of each of the other combinations.
The number of different colors that can be formed by the use of three colored lights, as shown in Fig. 1B, will depend upon the colors chosen. Experience has indicated that the colors red, blue, and green, when combined with each other in various proportions, will produce a wider range (or gamut) of colors than any other combination of three colors. Note, however, that if we used four different colors in our mixing process, we could produce an even greater number of different colors. With the addition of more and more colors to our mixing scheme, the reproducible range would widen somewhat. Obviously, however, a line must be drawn and the use of three colors has been standardized. The three colors chosen, red, green, and blue, are thus referred to as the "primary" colors although, as we shall see, the use of the word primary has been widely misinterpreted to mean that red, green, and blue will, in various combinations, reproduce all colors. This is only in a special instance.
The reason why three primaries were chosen, in preference to say four, probably stems from the belief that the eye behaves as though it contains three sets of nerves, with each set of nerves responsive to a different portion of the visible spectrum. Thus, one set of nerves has its greatest sensitivity in the blue region; another set is most sensitive in the green region; and the third set is most sensitive to red. Whether or not three sets of nerves actually exist has never been absolutely established. However, since the eye reacts as though such a condition does exist, it is reasonable to work on the assumption that it does.
The theory which serves to explain the ability of the human eye to distinguish various colors can also be employed to explain color blindness. In the eyes of a color-blind person. all of-the color sensitive nerves or retinal cones react in the same way to all colors. Hence, when colored light is viewed by these people, all three sets of nerves are similarly stimulated and the same result is obtained as though equal amounts of red, green, and blue light were intermixed. The color seen would be white, or some intermediate shade of grey. These people can distinguish between dark and light, but no more.
There are also people whose retinal cones differ sufficiently to see some of the colors, but not all. These people are known as partially color blind. Perhaps the best known instance of this is green and red color blindness. In the eyes of these people green or red appears grey. Fortunately, however, over 90 per-cent of the population have normal vision, which means that they are able to distinguish between all of the spectrum colors.
A diagram which is very convenient to use for color mixing is the tongue-shaped (or horseshoe-shaped) curve shown in Fig. 2. (Another name for this curve is chromaticity diagram.)
Around the perimeter of this curve are listed numbers that range from 400 at the lower left-hand corner to 740 at the farthest point to the right. These figures represent the wavelength of various spectrum colors in millimicrons. Thus, purple (violet) extends from approximately 400 to 450, blue extends from 450 to 500 millimicrons, green extends from 500 to 570 millimicrons, yellow extends from 570 to 590 millimicrons, orange extends from 590 to 610 millimicrons, and red extends from 610 to 740 millimicrons.
Any point not actually on the solid-line curve but within the diagram represents not a pure spectrum color but some mixture of spectrum colors. Since white is such a mixture, it, too, lies within this diagram; specifically, at point "C." This particular point was chosen at an international convention in England and is generally referred to as "illuminant C." Actually, of course, there is no specific white light, since sunlight, skylight, and daylight are all forms of white light and yet the components of each differ considerably. The color quality of a conventional black-and- vhite television receiver tube is represented by some point in the central region of the diagram about point "C."
The chromaticity chart lends itself readily to color mixing because a straight line joining any two points on the curve will indicate all the different color variations that can be obtained by combining these two colors additively. Thus, consider a line drawn connecting points "R" and "G" representing certain shades of red and green respectively. See Fig. 3. If there is more red light than green light, the exact point representing the new color will lie on the line, but be closer to "R" than "G." Point "R1" might be such a color. On the other hand, if a greater percentage of green light is employed, the color will still lie on the line connecting "R" and "G," but now, it will be closer to "G" than "R." Point "G1" might be such a color. This same line of reasoning can be carried out for any two colors on the chart.
(On the screen of a three-gun tri-color picture tube we can carry out the same experiment by turning off the blue gun and permitting only the electron beams from the green and red guns to reach the phosphor-dot screen. As one beam, say that from the red gun, is made more intense, the resultant color on the screen shifts closer to. red. On the other hand, if the red gun is turned down and the green gun beam is turned up, the resultant color takes on more and more of a greenish cast. When both guns are producing beams of equal intensity, yellow will be seen.)
Point "C," in the central region of this diagram, is taken to represent white or daylight. If we draw a line between point "C" and any point around the curve, we have a mixture of white light and a particular spectrum color. Thus, in Fig. 4, a line connects point "C" and green at 545 millimicrons (point "A"), indicating a mixture of white light and spectrum green. If the amount of white light is zero, then the pure spectrum green will be produced. As white light is added, the hue of the green changes and the point representing this mixture moves along the line toward point "C." We might consider this as diluting the green, causing it to become lighter and lighter.
(In a color tube, we dilute a solid color, say green, by adding more red and blue. The red and blue combines with some of the green to form white, thereby reducing the intensity or depth of the green.)
It is possible to specify the saturation of a color by its distance from point "C." Thus, consider point "B" in Fig. 4. This is half way between point "C" and point "A" and represents a mixture of green diluted 50 per-cent with white light. The saturation of the green at point "B" is 50 per-cent. Had the distance between point "C" and point "B" been 75 per-cent of the total distance between point "C" and point "A," we would have stated that the saturation of the color at point "B" was 75 per-cent. By moving point "B" closer and closer to the spectrum curve, its purity increases until it becomes 100 per-cent at the curve-point "A." By moving point "B" closer to point "C," its saturation decreases. At point "C," the saturation is said to be zero.
In connection with saturation, the word hue is frequently heard. Hue represents colors such as red, green, orange, etc. It is associated with color wavelength and when we label a certain color as green, or orange, or red, we are specifying its hue. Thus, hue refers to the basic color as it appears to us, while saturation tells us how deep the color is. If the color is highly saturated we say that it is a deep color, such as deep red, or deep green. If it contains a considerable amount of white light, we say it appears faded or pale, as a faded red or a pale green. Hue and saturation are psychological terms representing the observer's impression of a color and hence they can-not be defined as precisely as wavelength.
At the bottom end of the chromaticity curve, on the line drawn from deep blue to red, there is a series of colors which are combinations of red and blue in various proportions. These range from bluish purple to purplish red. It can be seen that this line completes the curve of Fig. 2. However, this line should not be considered in the same sense as the rest of the curve. It does not contain any spectrum colors but only combinations obtained from mixing spectrum colors. Because of this, the region at the back end of this tongue-shaped curve is known as the region of non-spectral colors. The boundaries of this region are obtained by drawing dotted lines from point "C" to red at 700 millimicrons and from point "C" to blue at 450 millimicrons. The remainder of the diagram above these dotted lines is known as the region of spectral colors. The entire diagram is known as the domain of real colors.
One further term used in connection with this diagram is complementary color. Any two colors which can by themselves form white are known as complementary colors. Thus, in Fig. 5, the line connecting point "F" with point "G" passes through point "C" and hence, the colors at "F" and "G" are said to be complementary to each other.
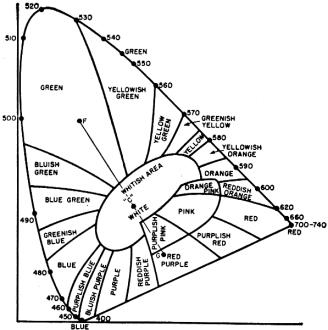
We have previously seen that a line drawn between two points representing two different colors contains all of the combinations that can be derived using those two colors. If, now, we wish to determine what range or gamut of colors can be obtained from any three given colors (say "R1," "G1," and "B1"), we would draw connecting lines to each of the colors. See Fig. 6. The result is a triangle. We can produce any color within this triangle by various combinations of the three colors, URt," "G1," and HB]."
The. wavelengths of "R1," "G1," and "B1" chosen for television fall near 610 millimicrons for the red, near 540 millimicrons for the green, and near 470 millimicrons for the blue. These are actually the values used for the triangle drawn in Fig. 6 and by studying this diagram you can see the extent of the color range obtainable on a color television receiver. Note that colors not included within the triangle will not be reproduced by any combination of the three primary colors chosen.
This, of course, brings us back again to the statement made previously, that three primary colors cannot reproduce all colors simply by adding the three primary colors together. In color television, of course, only those colors which can be produced by adding the primaries together can be considered, this being the only practical approach possible.
The choice of suitable primary colors for television depends principally upon what type of color phosphors can be obtained for the receiver picture tube. Originally it was felt that the color picture would be traced out on a black-and-white screen and then the light passed through a color filter to present the observer with the "color" image. This was the method employed in the CBS system and in the early forms of the RCA system. However, with the development of a color tube, phosphors are employed which emit colored light directly, leading to a less cumbersome system physically and a more efficient system optically.
A considerable amount of research work is being done on evolving phosphors which will provide as wide a gamut of colors as possible. In recent tubes, a willemite phosphor (Zn2 SiO4: Mn) was used for the green, a sulphide phosphor (ZnS : Ag : MgO) for the blue, and a third phosphor, Zn3 (PO4)2 : Mn for the red. These primaries provide a fairly wide range of colors, as seen in Fig. 6. By comparison, the area covered by printing inks is much smaller. It may be that as the art advances, the color range of the phosphors will be extended, although the colors now obtainable are wholly satisfactory.
When the NTSC system of color television was under development, a considerable amount of research was done on how much color the average human eye really sees. This work, in conjunction with other data which has appeared from time to time, brought forth several very interesting facts.
1. The theory that vision is a three-color process is true only when the object viewed is relatively large. On a television screen, this refers to objects which are produced by video frequencies from 0 to .5 mc.
2 For medium-sized objects, say those produced by .5 mc. to 1.5 mc. video frequencies on a television screen, only two primary colors are needed. Blues and yellows are among the first colors to lose their color and become indistinguishable from gray within this range.
3. For very fine detail, say those reproduced by video frequencies from 1.5 to 4.0 mc., all people with normal vision are color blind. In other words, all that is seen are shades of brightness.
The conclusion to be drawn from the foregoing is that 4 mc. color is not necessary. All we require is color up to 1.5 mc. And even within this range, we need all three colors only to .5 mc. and only two primaries for the color signal extending from .5 mc. to 1.5 mc. In the formation of the NTSC signal, these facts were put to use by employing one color signal, called the "Q" signal, with a range from 0 to .5 mc. and a second color signal, called the "I" signal, with a bandpass from 0 to 1.5 mc. The rest of the video picture, containing all of the fine detail, is reproduced in black-and-white by a monochrome signal and the eye is none the wiser. As a matter of fact, a full color television signal consists of a 0 to 4 mc. monochrome video signal (just as we have in black-and-white broadcasting) plus a color subcarrier containing the "I" and "Q" color signals mentioned previously. It has been truly said that the NTSC system is a "colored" television system.
The monochrome signal possesses such alternate names as brightness signal and luminance signal. The color portion is frequently referred to as the chrominance signal.
In the next article of this series we will see how the NTSC color signal is formed and of what it consists.
(To be continued)
Color and Monochrome (B&W) Television Articles
- Color Television Has a Problem - May 1966 Radio-Electronics
- What You Want to Know About Television - February 1949 Popular Science
- Electronic Color Television Is Here - February 1947 Popular Science
- Television on the Job - February 1947 Popular Science
- What You Should Know About X-Ray Radiation in TV Sets - November 1967 Radio-Electronics
- Apollo 10 & 11 Color Video Camera Used Rotating Color Wheel - August 1969 Radio-Electronics
- What Happened at Oslo? - ITU Color TV - January 1967 Radio-Electronics
- Color Television Past Present Future - January 1954 Radio-Electronics
- Basic Color TV, Part I - January 1954 Radio-Electronics
- How Television Cathode Ray Tubes Are Made - July 1938 Radio News
- Color Television Fundamentals, Part 1 - March 1954 Radio & Television News
- Color Television Systems - January 1951 Radio-Electronics
- Simulating a CBS Color Signal - September 1951 Radio & Television News
- The Strange World of Color Vision - January 1958 Radio Electronics
- Color Television? - December 1949 Radio & Television News
- Fundamentals of Color TV: The NTSC System - April 1954 Radio & Television News
- Color TV - October 1953 Radio & Television News
- The Color TV X-Ray Problem - November 1968 Radio-Electronics
- Introduction to Modern Television - February 1939 Radio News
- Television Industry Prepares for Postwar - January 1945 Radio News
- Television Transmission Lines - May 1952 Radio & Television News
- Television in Great Britain - January 1945 Radio News
- Television Set Shipments by Areas - May 1949 Radio & Television News
- Television Forges Ahead - March 1930 Radio News
- The Television Test Pattern - January 1949 Radio & Television News
- Television Tubes by the Thousands - December 1947 Radio News
- Television I.Q. Quiz - October 1948 Radio & Television News
- Television, As I See It - January 1945 Radio News
- The Outlook for Color TV - March 1957 Radio & Television News
- Color TV Today - September 1959 Popular Electronics
- Mac's Service Shop: Motorola's 1974 Color TV Receivers - November 1973 Popular Electronics
- Mac's Service Shop: Cost of Color TV Service - February 1969 Electronics World
- Practical Color TV for the Technician - September 1957 Radio & TV News
- Anxiety Amid Affluence: Why Color-TV Makers Worry - December 27, 1965 Electronics Magazine
- Fundamentals of Color TV - Tri-Gun Receiver Circuits - June 1954 Radio & Television News
- 3-D Color TV for the Future? - May 1958 Radio News
- Mac's Radio Service Shop: Barney Takes on Color - February 1955 Radio & Television News
- TV X-Rays - April 1970 Radio-Electronics
- TV X-Rays Are Back - June 1969 Radio-Electronics
- Television in Twelve Colors - October 1930 Radio-Craft
- Trinitron - Still a Mystery? - February 1972 Popular Electronics
Posted January 26, 2021

Copyright: 1996 - 2026 |
About RF Cafe RF Cafe began life in 1996 as "RF Tools" in an AOL screen name web space totaling 2 MB. Its primary purpose was to provide me with ready access to commonly needed formulas and reference material while performing my work as an RF system and circuit design engineer. The World Wide Web (Internet) was largely an unknown entity at the time and bandwidth was a scarce commodity. Dial-up modems blazed along at 14.4 kbps while tying up your telephone line, and a lady's voice announced "You've Got Mail" when a new message arrived... |
Copyright 1996 - 2026 All trademarks, copyrights, patents, and other rights of ownership to images
and text used on the RF Cafe website are hereby acknowledge My Hobby Website: My Daughter's Website: |